St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Babis Kalodimos Lab
Investigating the conformational landscape of protein kinases and molecular chaperones
About the Kalodimos Lab
Protein kinases and molecular chaperones are some of the most challenging biological systems involved in disease. They play central roles in pediatric cancer and other catastrophic disorders of childhood. Our work centers on understanding the structure and dynamics of these complex protein machineries. We combine high-resolution NMR spectroscopy with other structural approaches to decipher normal and pathological mechanisms of action.

Our research summary
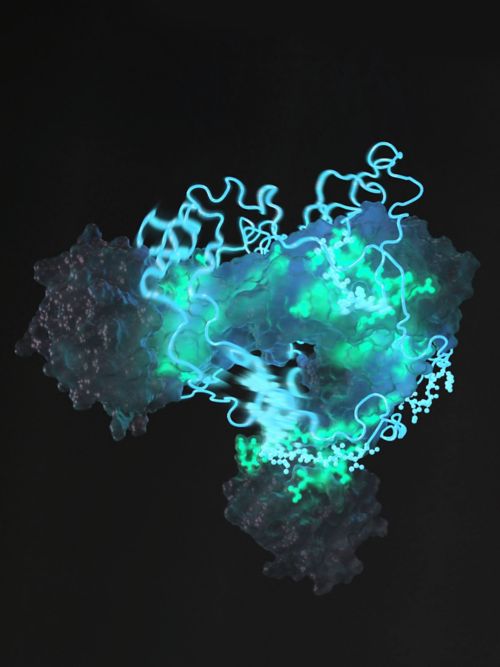
Our laboratory aims to understand the structure and dynamics of the complex protein machineries that underlie key biological processes in the body. Our research efforts focus on two major biological families that are involved in disease — protein kinases and molecular chaperones. Leveraging state-of-the-art technologies in NMR spectroscopy, X-ray crystallography and cryo-EM, the projects in our laboratory revolve around how protein kinases and molecular chaperones function, both in vitro and in vivo.
Protein Kinases
Involved in important processes both inside and outside of the cell, protein kinases are the most frequently mutated domains in cancer and are therefore often targeted for therapeutic intervention. In our protein kinase research, we explore – on an atomic level – the rare conformational states that kinases transiently adopt as it functions. These states have historically been difficult, if not impossible, to capture and visualize but our laboratory recently employed state-of-the-art NMR spectroscopy techniques to show the different active and inactive conformations of the Abl kinase. We are now extending this novel approach to other kinases involved in disease.
The ultimate goal of this area of research is to better understand how the activity of protein kinases is regulated, since out-of-control kinase activity often leads to tumorigenesis, cancer, inflammation, and neurological diseases. In addition, our laboratory aims to understand the mechanisms of disease, leveraging the data we generate to identify selective inhibitors for therapeutic intervention and gain insight into drug resistance.
Mechanism of ligand-induced dimerization and activation of ALK. Animation By: Zhaowen Luo
Molecular Chaperones
In our molecular chaperone research, we are exploring large and dynamic complexes and the proteins that are upregulated when there are many unfolded or misfolded proteins in the cell. Molecular chaperones represent the first line of defense by working to prevent misfolding or aggregation but the interaction of chaperones with these unfolded or misfolded substrates is transient in nature.
Our laboratory has spearheaded novel approaches that use NMR spectroscopy and other techniques to observe the molecular chaperones with their unfolded proteins. The technique we pioneered allows us to develop high-resolution structures and to observe how chaperones recognize and connect with proteins. Armed with a more in-depth understanding of what is happening within the cell between molecular chaperones and proteins, we aim to apply this line of investigation to other systems and develop new therapeutic interventions.
Selected Publications
Contact us
Babis Kalodimos, PhD
Structural Biology MS311
St. Jude Children's Research Hospital
Follow Us

Memphis, TN, 38105-3678 USA GET DIRECTIONS