St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Stacey Ogden Lab
Understanding cell to cell communication by deciphering mechanisms of Sonic Hedgehog signaling in development and disease
About the Ogden Lab
The Sonic Hedgehog (SHH) signaling protein plays an important role in normal development and tissue homeostasis. Corruption of the pathway leads to neurodevelopmental disorders and a host of cancers. Our laboratory seeks to define the regulatory processes governing SHH pathway activity during development. An improved understanding of how the SHH pathway works in healthy systems will illuminate mechanisms of dysfunction and how it might be targeted to prevent or treat diseases.
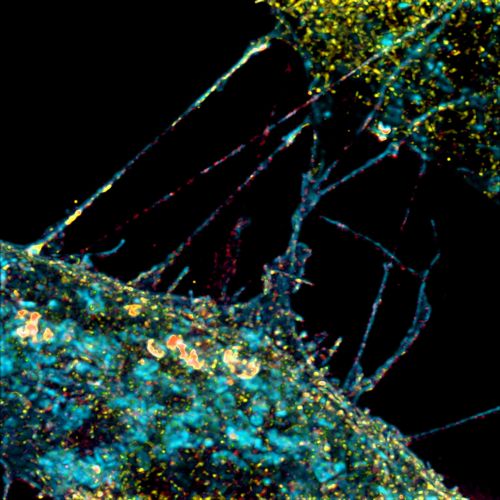
Our research summary
SHH protein is produced in and released from cellular organizing centers during tissue patterning to instruct fate decisions in neighboring cells. Our laboratory focuses on pivotal components of the SHH signal transduction cascade. We are working to define 1) how SHH signals are activated and guided through distinct effector routes 2) how the amplitude and duration of the transcriptional response is controlled and 3) how cells involved in SHH signaling communicate through cytonemes.
Noncanonical SHH Signaling
SHH signal is transduced across the cell membrane to intracellular effectors by the G protein-coupled receptor Smoothened (SMO). Our lab was the first to provide evidence that SMO can bias its signal between canonical and noncanonical effector routes. The molecular mechanisms driving non-canonical SMO signaling and how it functions in development are unexplored. We have developed a novel SMO mutant model to dissect the contribution of non-canonical SMO signaling to tissue patterning during development.

Regulation of the SHH Transcriptional Response
SHH-mediated SMO activation instructs gene expression through GLI effectors which can either function as activators or repressors. Our laboratory provided the first evidence that GLI could be partitioned into discrete complexes within the nucleus, a crucial mechanism by which GLI activator is balanced to shape transcriptional response. We are currently exploring how GLI assemblies are controlled. An understanding of how to manipulate GLI assembly formation may reveal therapeutic alternatives in cases where current SHH pathway inhibitors are insufficient.

SHH Communication
SHH signals are transported within and between cells to provide instructional cues for development and homeostasis. Cytonemes are specialized filopodia that help establish and reinforce the SHH morphogen gradient, facilitating a graded signaling response across developing tissues. Our lab has developed and validated cell culture and fixation and imaging protocols capable of preserving cytonemes for advanced imaging and functional analysis. We are continuing to build the protocols, methodologies, and reagents to observe these processes in vivo. We are exploring whether defects in cytoneme dynamics contribute to SHH-associated disorders.
The research in our laboratory uses a combination of biochemistry, cell biology, and genetic model systems, as well as bioinformatics and imaging techniques including electron-, confocal-, and stimulated emission depletion (STED)- microscopy. We have established robust collaborations across the department and the institution.

Selected Publications
Contact us
Stacey K. Ogden, PhD
Member, St. Jude Faculty
Departmebnt of Cell and Molecular Biology
MS340
St. Jude Children’s Research Hospital
Join us
The Ogden Lab is accepting applications for a Senior Scientist of Lab Research

Memphis, TN, 38105-3678 USA GET DIRECTIONS