St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Shared Resource Spotlight: Cell and Tissue Imaging Center
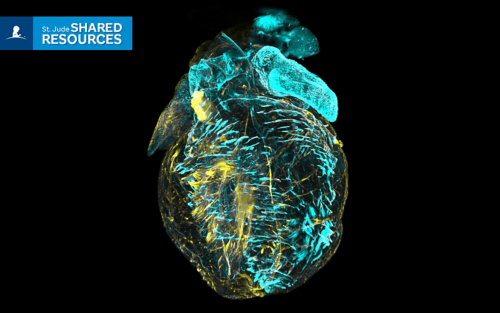
Experts at the St. Jude Cell and Tissue Imaging Center can help visualize details within tissues, such as this image of a mouse heart taken after organ clearing and immunostaining. Courtesy of Aaron Taylor.
As early as 1665, scientists used imaging to understand the world. From those early hand-drawn pictures of objects under a microscope, today’s technology can create images of complex 3D tissues due to the ways that imaging technology has adapted and advanced.
In the 1990s, it was cutting-edge to inject dyes like trypan blue in blood vessels to see them more clearly. Today, technology has advanced to allow for organ clearing. This technique makes biological samples transparent, enabling whole-body fluorescent imaging to visualize the intricate network of multiple cell lineages that comprise the entire organism. It is an example of powerful ways technology can allow researchers to peer into the biology of living things.
With the importance of imaging well established across scientific disciplines, the ability to conduct these types of experiments and analyses is essential to the St. Jude research enterprise. Various imaging technologies are readily available to all investigators at St. Jude through Shared Resources — centralized innovation hubs.
Shared Resources at St. Jude are at the heart of scientific discoveries. Ranging from vector development, protein isolation, new target discovery and disease diagnosis to clinical translation of new anti-cancer therapeutics, the support of shared resources plays an instrumental role in every laboratory’s scientific story. Technological advancements in imaging have allowed researchers to visualize tissues at molecular and genetic levels and trace disease development. The Cell and Tissue Imaging Center shared resource plays a vital role in detecting the cellular architecture of an organism or identifying if the structure has collapsed when struck by disease at a cellular or atomic level.
Cell and Tissue Imaging Center
The efforts of the Cell and Tissue Imaging Center Light Microscopy (CTIC-LM) and Electron Microscopy (CTIC-EM) shared resources are led by research operations directors Aaron Taylor and Cam Robinson, PhD, respectively. These resources focus on making high-speed and high-resolution imaging technologies available to scientists at St. Jude. CTIC staff support a wide range of sample preparation, imaging and analysis technologies that are a fundamental piece of what makes discoveries possible.
Advanced technology for light microscopy
As microscopy technologies evolve, we can examine where proteins are located within a cell or study the distribution of cell types across complex tissue. Traditionally, visible wavelengths shone and detected with microscopes have been used to image fixed samples. However, this design is not light efficient — the entire sample is illuminated for each 2D image obtained — and visible wavelengths are prone to absorption and scattering, necessitating high laser powers to generate useful images. As a result, conventional approaches translate poorly to the problem of 3D imaging of living samples.
“Multiphoton microscopy uses infrared (invisible) light to reduce phototoxicity and laser power but only acquires one point (pixel) at a time, making a volume of millions of points arduous to acquire,” said Taylor. “Light sheet microscopy uses one (or more) objectives to illuminate only a thin plane in the sample. At the same time, a separate objective collects the emissions using a camera. This design is both fast and light efficient.”
“These benefits can also be extended to sub-cellular volumetric imaging,” Taylor added.
In the St. Jude CTIC-LM, researchers can employ lattice light sheet microscopy to image the entirety of living cells across thousands of time points. This duration was previously not possible.
“CTIC-LM aspires to drive and accelerate the use of advanced imaging across St. Jude,” Taylor said. “The Center offers best-in-class microscopes encompassing virtually every commercialized fluorescence imaging modality.”
For example, the Center’s new Zeiss lattice light sheet microscope enables volumetric imaging of living cells over thousands of time points with minimal phototoxicity. The Center also validates technology pipelines for expansion microscopy, tissue clearing and multiplexed imaging. This enables researchers to leverage these techniques without spending significant effort troubleshooting independently.
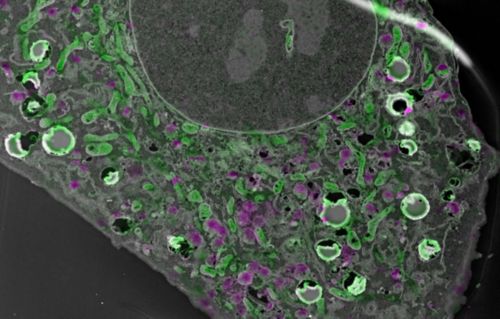
Correlative Light and Electron Microscopy (CLEM) generates images such as this, where imaging of embedded fluorescent proteins and EM tomography are combined. Courtesy of Aaron Taylor.
Electron microscopy gives a different view
Electron microscopy helps researchers zoom into cellular structures, dissecting them into organelles and protein structures at an atomic level. The technology used in electron microscopy provides structural details for different biomolecules at a high resolution and allows researchers to understand cellular health and dysfunction.
Electron microscopy technology has been evolving for over a hundred years. Originally, researchers used transmission electron microscopy to observe the ultrastructure of small molecules such as carbon black at a high resolution. Over time, scanning electron microscopy was introduced to detect the topography of thicker 3D samples and identify simple structures that make up complex molecules.
Today, cryopreservation allows for maintaining the tissues at temperatures below freezing to preserve the structure of the samples. Newer technologies involve imaging cryopreserved samples to observe the complex macromolecular structure at a molecular level. Moreover, a technology called correlative light and electron microscopy combines electron microscopy with light microscopy to visualize the morphology and fluorescently label the target of interest in the same section.
“CTIC-EM is working to enhance correlative imaging offerings with advanced microscope systems,” said Robinson. “We are also integrating cryogenic sample preparation methods to improve ultrastructural preservation while expanding the ability to apply different labeling technologies within the correlative imaging space.”
CTIC shared resource enables future discoveries
Shared resources at St. Jude are unparalleled, providing support and expertise to researchers to assist with one-on-one consultations on protocol development, data curation and image analysis. The way scientists visualize the world around us and within us has changed tremendously as technology has made it possible to look further and deeper into the biology of living things. This most fundamental aspect of scientific discovery is thriving at St. Jude, largely due to the dedicated staff and advanced technologies available in the CTIC.






