St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Making innovative HIV medications an option for younger ages

By participating in clinical trials like GS5578, St. Jude is working to make innovative HIV medications an option for younger ages.
Do you remember to take your medicine every day? Or do you hesitate, perhaps even forgetting, because you do not like how it tastes or are overwhelmed by the size or number of pills to swallow? How do your feelings about medicine change when needles are involved? If you need help taking medicine, is the medicine easy for others to prepare for you?
These are all important considerations to make when deciding what medication options are best. However, for children, especially very young children, these options are limited, making the administration of medication a challenge.
Having a variety of medicines available that offer different administration options — how and when these medications are taken — can make all the difference when it comes to children receiving the medicines they need, when they need them. Having a variety of administration options gains importance when preventing or managing chronic illnesses that require lifelong treatment, such as the human immunodeficiency virus (HIV). To prevent or treat HIV, patients must consistently take antiretrovirals to either prevent a viral exposure from progressing to infection or to bring the virus levels to an undetectable level.
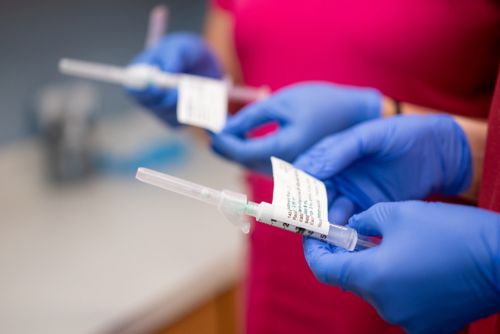
Clinical staff at St. Jude hold prepared injections of cabotegravir and rilpivirine.
While a variety of medication options exist that accommodate the administration preferences of adults and adolescents living with or trying to prevent HIV, those options are fewer in younger age groups. St. Jude is working to change this, though. Through participation in multisite clinical trials, St. Jude health care professionals are helping expand existing HIV treatment and prevention options to all ages.
The long and not short of evaluating medication options in younger ages
Bringing treatments established in adults to younger age groups is a complex process. When a new treatment becomes available, its safety and efficacy are first assessed in adults and subsequently approved by the Food and Drug Administration (FDA) for use only in adults. To make these innovations in care available to younger individuals, pediatric clinical trials must be conducted to determine safety, dose, and how the drug distributes and metabolizes in the body (called pharmacokinetics), for different ages and weights, often descending from the oldest to the youngest age groups.

Pharmacy technician Angela London (left) and clinical pharmacist Susan Carr, PharmD, (right) prepare the dissolvable HIV medication (B/F/TAF) being tested on GS5578.
Conducting separate trials means there can be years between when a drug is approved for adults and when it becomes available for adolescents, school-age children, young children, toddlers, infants and newborns.
IMPAACT 2017 to IMPAACT 2036: Expanding injectable options
In 2021, a long-acting injectable medication was approved for adults living with HIV. The next year, the NIH-funded International Maternal Pediatric Adolescent AIDS Clinical Trials Network (IMPAACT) 2017 study, also referred to as the More Options for Children and Adolescents (MOCHA) study, facilitated the approval of long-acting injectable antiretrovirals, cabotegravir and rilpivirine, administered in combination via an injection once every two months, for adolescents aged 12 to 18. Four years later, clinics across the world are using the pharmacokinetic and safety data from that trial to inform the dose planning for younger children aged 2 to 11 who wish to explore a similar regimen as part of the multi-site clinical trial, IMPAACT 2036, operated by the global IMPAACT clinical trial network.
When designing and testing new treatments, considering the preferences and fears of children and their caregivers remains top of mind for investigators. Aditya Gaur, MD, Division of HIV and Translational Medicine director, Department of Infectious Diseases member, said that “one cannot appreciate enough the contributions of the children and their families who participate in these clinical trials. In clinical research, it is critical that participants are fully informed and every effort is made to inform both the care provider and the child about the study in an age-appropriate way. These children and their families are truly the ones that help bring the possibility of exciting new treatment options to pediatrics.”

Angela London and Susan Carr, PharmD, give a prepared dose of B/F/TAF to Lauren Braskich, RN, and Kierra McCallum, RN, nurses who will administer the medication to patients enrolled on GS5578.
St. Jude was the first site in the world to enroll a patient and initiate the long-acting study medications on the IMPAACT 2036 study.
As a clinical pharmacist who works on every standard-of-care treatment and experimental protocol implemented in the HIV clinic at St. Jude, Susan Carr, PharmD, Department of Pharmacy & Pharmaceutical Sciences, reflects on the possibilities of IMPAACT 2036. “We’ve seen and heard in the adult population how much their lives have improved with the long-term injectable antiretroviral option simply because they are not having to take medicine every day. Now, we want to give the kids that same option and chance for a quality-of-life improvement,” she said.
Empowering patients and caregivers to explore medication options that consider their preferences helps ensure that taking medicine is not a stressor hindering care or quality of life.
Bringing a new prevention option to the youngest age group
In young infants, antiretrovirals are mostly used for HIV prevention rather than treatment. Antiretrovirals reduce the likelihood of viral transfer during pregnancy or around the time of delivery if the pregnant person is living with HIV. These prevention efforts commence as soon as the pregnant person learns their HIV status; HIV tests are administered to pregnant people in the first and third trimesters as part of their prenatal checkups. Consistent treatment of the pregnant person ensures viral load levels drop to and remain at an undetectable level, which reduces the likelihood of viral transmission.
Newborns exposed to HIV receive antiretroviral treatment beginning shortly after birth to prevent any possible viral exposure from progressing to infection. Infants at highest risk of HIV infection — for example, if the birth parent had a high level of HIV at delivery — are given combination antiretroviral therapy after birth. For these infants, even if preventative measures were not successful, antiretroviral treatment for the newborn has already been started.

Chloe Zorbino, RN, stands outside a clinic room in the HIV clinic at St. Jude.
Currently, combination antiretroviral therapy for infants requires three separate medications, each of which must be given twice a day. While there are several fixed-dose formulations allowing a single daily administration of antiretroviral therapy for adults and older children, there are currently no such options for infants.
GS5578 is the first study in newborns to evaluate a multidrug fixed-dose formulation with a combination of the antiretroviral medications bictegravir, emtricitabine and tenofovir alafenamide (B/F/TAF), which is a pill dissolved in liquid and administered via an oral syringe. St. Jude was the first site globally to enroll in this study.
Babies whose caregivers enrolled on GS5578 will receive one dose of B/F/TAF five days after birth and then another 10-15 days later and will be followed in the study until eight weeks of age. The study will assess how well the babies tolerate the medicine, how safe it is and the pharmacokinetics of B/F/TAF.
If this study determines safe and effective dosing for B/F/TAF in newborns, the study sponsor would then seek FDA approval for this combination drug in infants. A multidrug fixed-dose formulation would decrease the likelihood of dosing errors and streamline and optimize the medication administration process to ensure an improved experience for both baby and caregiver.
Katherine Knapp, MD, Perinatal HIV Program medical director, Department of Infectious Diseases associate member, emphasizes B/F/TAF is revolutionary for this age group, especially for the caregivers who must administer the medication.
As medical director of the Perinatal HIV Program at St. Jude, Knapp assesses and provides care to all the children born to people living with HIV in Memphis, where St. Jude is located, and the surrounding areas. She sees approximately 35-45 babies born in the area per year and has heard first-hand the challenges these new parents face. Such challenges include trying to give medicine to babies who inherently resist and adhering to a demanding medication schedule that calls for multiple doses per day, all combined with the fatigue of caring for a newborn.
“This medication option is going to be a game changer for the littlest ones because we currently have very few options to use with them, which makes it a challenge for both baby and caregiver. We really need an easier-to-administer regimen that’s well tolerated, and the one we’re testing on GS5578 is that. It’s a very efficacious drug, and expectant parents are eager to participate in the trial,” said Knapp.
Considering what motivates caregivers to enroll children in clinical studies
Investigators do not take this eagerness to participate in clinical research lightly. Gaur emphasizes the importance of considering the motivations behind why caregivers may enroll themselves or their children in clinical studies, whether that be a benefit to the self or a future benefit to others.

Katherine Knapp, MD, and Aditya Gaur, MD, discuss the new HIV treatment options being assessed at St. Jude for the youngest ages.
When approaching patients and caregivers about the possibility of participating in a clinical trial such as IMPAACT 2036, trial coordinators — the individuals responsible for the regulatory components, documentation, training compliance, and information provision to patients and caregivers — consider much more than whether the patients and caregivers meet the initial criteria for participation in the trial.
For these two trials, the participants are usually existing St. Jude patients, so the established relationships between care providers and the patients and/or caregivers play a vital role in how participants experience clinical research at the institution.
For Chloe Zorbino, RN, clinical research associate-registered nurse (CRA-RN), Department of Infectious Diseases, these relationships provide the foundation necessary for patients to feel respected and cared for as part of the clinical research process. As the CRA-RN for GS5578, Zorbino builds relationships with the patients and caregivers she regularly takes care of in the HIV Clinic at St. Jude. If the circumstances are right and the patients or caregivers choose to enroll in a clinical trial, Zorbino walks with them through all stages of the study.
“Giving your brand-new baby an investigational drug is a big ask,” she said. “It’s a lot to ask of new moms, so the relationships we build with caregivers are the foundation that we build their experience of the study on.”
The trial coordinator for IMPAACT 2036, who works in the Department of Infectious Diseases, explained that a motivating factor for enrollment in this particular trial is the fact that taking a pill every day can interfere with the developing social lives of young children. The necessity of a daily pill makes it difficult to honor the private nature of taking medicine, especially when young children may not be ready to assume responsibility for their medication and need help from adults or are afraid to take their medicine around others due to the stigma of HIV.
The trial coordinator said, “You have to weigh all the benefits and challenges of each medication, which is why it’s so important to have many options available; it allows the patient and caregivers to decide what is best for them — they have the power to decide; it is not for us to decide for them.”
However, those decisions are often not motivated solely by interest in how clinical trial participation can benefit them.
Carr, who helped educate the first caregiver enrolled on GS5578 at St. Jude on how to administer B/F/TAF to her newborn, recalls how proud the caregiver was to be the first person to administer the medicine to her baby as part of the study. “She just wanted to give back and not only help her baby, but future babies. That is the resounding thing I hear in investigational studies across St. Jude, whether it’s infant caregivers or older kids or adults: They want to help others. There is an amazing community of patients at St. Jude who want to give back [by participating in clinical research].”






