St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
St. Jude Family of Websites
Explore our cutting edge research, world-class patient care, career opportunities and more.
St. Jude Children's Research Hospital Home

- Fundraising
Simulations help create real-world solutions to global health challenges
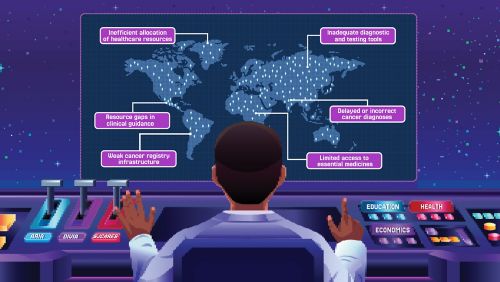
The St. Jude Global Disease Burden and Simulation Unit is a multidisciplinary program that uses data and innovative tools to help improve childhood cancer care worldwide.
Decisions about childhood cancer are often made with inadequate or inequitable evidence because that is all that is available. Around the world, particularly in resource-limited countries, clinicians, policymakers and health systems face critical gaps in quantifying how many children have cancer, which forms of the disease are most prevalent and what treatments are most effective. Without reliable information, resources can be misallocated, families may bear disproportionate financial burdens and survival outcomes can vary dramatically depending on where a child is born.
The Global Disease Burden and Simulation (DBS) Unit at St. Jude is working to change that. By generating high-quality and standardized data, developing innovative modeling tools and collaborating with stakeholders worldwide, the DBS Unit helps provide the evidence needed to guide decisions that improve outcomes for children with cancer around the world.
“When we started the DBS Unit, the data that existed was either inaccurate or simply didn’t exist,” explained Nickhill Bhakta, MD, MPH, St. Jude Global DBS Unit director. “There was no reliable data to reference.”
Global pediatric cancer burden and surveillance
Early on, the DBS Unit began working to improve the accuracy and utility of childhood cancer data. Collaborations with the Institute for Health Metrics and Evaluation (IHME) helped refine global disease burden estimates, leading to a publication in The Lancet that classified over 90% of childhood and adolescent cancers. This provided policymakers and health systems with accurate data to guide funding, interventions and strategies to shape cancer care in country-specific contexts.
The unit has led the St. Jude partnership with the International Agency for Research on Cancer (IARC) to support population-based cancer registries. Together, St. Jude and IARC have developed the Global Initiative for Cancer Registry Development (ChildGICR) to address the global gap in cancer epidemiologic data. This initiative focuses on research, education and implementation to increase childhood cancer registration globally and thus the quality of the data.
Additionally, the unit works with the CONCORD Programme, which produces global survival trends from cancer registry data. Historically focused on adults, CONCORD has expanded to include pediatric cancers in its most recent iteration. Advocacy and collaboration from St. Jude helped highlight the importance of including childhood cancers, ensuring that children are no longer overlooked in global survival estimates.
Collectively, these efforts have strengthened the accuracy and utility of childhood cancer data, providing policymakers and health systems with the evidence necessary to design effective pediatric cancer care programs and monitor progress.
Creating hospital registries to improve care with SJCARES
Less than 12% of the world’s children are covered by population-based registries, leaving most countries without reliable data. Recognizing this gap, the DBS Unit is leading an effort focused on supporting the creation of hospital registries, which often serve as de facto population registries in countries where there are only one or two hospitals that treat children with cancer.

Nickhill Bhakta, MD, MPH (L), Global Disease Burden and Simulation Unit director, Department of Global Pediatric Medicine associate member, Nancy Bolous, MD, MA, MSc (R), Global Disease Burden and Simulation Unit lead researcher, Department of Global Pediatric Medicine assistant member, and their teams drive the work of the Global DBS Unit, improving childhood cancer care worldwide through data, innovative tools and health economics research.
In 2020, the DBS Unit launched the SJCARES Registry Tool, a hospital-based pediatric cancer registration and reporting system, to directly generate high-quality, interoperable data from hospitals. The registry establishes standardized data elements, tools, educational resources and quality monitoring systems, enabling hospitals around the world to collect and share consistent, actionable data.
Since its launch, SJCARES has grown to include a cloud-based network consisting of over 30,000 patients across 60 hospitals, with an additional 50 hospitals using the same data elements but with local data management. The registry integrates with visualization platforms, allowing real-time insights into trends, outcomes and treatment effectiveness with the primary aim of supporting hospitals in identifying and tracking opportunities and gaps. With this infrastructure, the DBS Unit can support quality improvement initiatives and help refine global childhood cancer burden estimates
From data to action
Insights gained from the DBS Unit’s simulations and data modeling have provided a foundation for creating practical tools to directly improve patient care worldwide. One key outcome of this approach is the Adapted Resource and Implementation Application (ARIA) program. The ARIA Guide was developed to create globally applicable, evidence-based guidelines for the treatment of complex childhood cancers that can be applied regardless of a child’s location or available resources. For example, a hospital in Zimbabwe may have different medications available than a hospital in Nepal or a hospital in the United States.
Unlike traditional clinical guidelines, which often focus on single, simple diseases and are limited to a country or continent, the ARIA Guide addresses the full spectrum of pediatric cancers. The program integrates surgery, radiation, multiple chemotherapy regimens and the management of treatment-related complications to provide treatment guides that can work within the context of different regional settings.
The ARIA Guide addresses real-world challenges in cancer care, such as limited access to diagnostic imaging equipment, radiation or surgery, by providing clear treatment guidance adaptable for any clinical setting. Its guidelines are integrated into a decision support tool enabling healthcare providers to quickly identify the most appropriate course of treatment for each patient.
The ultimate goal of the ARIA Guide is to create a fully integrated suite of tools that combine guidelines, real-world data and diagnostics to improve childhood cancer care globally.
Transforming diagnostics
But tailoring appropriate treatment strategies is only part of the challenge of providing pediatric cancer care. To start, cancers need to be accurately diagnosed, but there are persistent gaps in diagnostic capacity, particularly in low-resource settings. Data from the DBS unit has shown that nearly 50% of children globally who develop cancer are never diagnosed. Furthermore, even among those who get a diagnosis, another 50% of children experience significant, potentially life-threatening delays in diagnosis. To help, the DBS Unit, together with St. Jude researchers in the Department of Pathology, formed the Diagnostic Innovations using Value-based implementation models to Increase Access (DIVIA) program.
DIVIA is an institutional initiative designed to address a critical gap in diagnosing childhood cancers in low-resource settings, where advanced laboratory tools for pathology or molecular diagnostics are often unavailable. The multiple tests, specialized technicians and complex supply chains that pose barriers to diagnosis are difficult for many health systems to manage.
“We started this journey by asking ourselves, ‘Why can’t we utilize next-generation sequencing tools to simplify leukemia diagnosis?’” explained Bhakta. “We realized we could not spend 20 years trying to replicate what we do at St. Jude and instead needed to leapfrog conventional diagnostics. It was this frame of thinking that led to the creation of DIVIA.”
DIVIA is designed to expand access to advanced cancer testing through cost-aware molecular diagnostics, thus simplifying the identification of childhood cancers. Much of the team’s early work focused on evaluating whether laboratories in resource-limited settings could generate data of sufficient quality to diagnose children with cancer. During the COVID-19 pandemic, there was a proliferation of genomic data from around the world, but much of that data was of varying quality.
The team published a key breakthrough in Leukemia showing that rapid, cost-effective genomic classification of acute leukemias was feasible. Using a novel “adaptive DNA-based” assay, researchers from St. Jude and UNC demonstrated the potential to generate comprehensive genomic analyses from a single sample. Within 24 hours, they identified gene fusions, copy number alterations, chromosome-level abnormalities, single nucleotide polymorphisms and pharmacogenomic insights (how drugs work in the context of certain mutations), at a fraction of the cost of conventional methods.
“This is a human story with human impact,” emphasized Bhakta. “For example, in Pakistan, before DIVIA, they only had karyotyping, flow cytometry and a couple of polymerase chain reaction (PCR) tests. Now, with comprehensive risk-stratification and pharmacogenomic data from a single assay, clinicians can identify which patients may benefit from reduced chemotherapy doses, lowering the risk of toxic death while also saving money.”
Pilot implementation studies placing these tools as routine clinical tests across hospitals in Pakistan, India, the Netherlands and the U.S. have shown that DIVIA can deliver high-quality results comparable to diagnostic methods considered the “gold standard.” Based on these promising initial results, the DIVIA program was selected as a 2025 recipient of the Amazon Web Services Imagine Grant Children’s Health Innovation Award and recently partnered with Oxford Nanopore Technologies (ONT) to provide free equipment and discounted flow cells, small chambers that hold DNA during sequencing, to 11 St. Jude Global Alliance sites to evaluate sustainable scale-up mechanisms worldwide.
Finally, the DIVIA team is now actively evaluating innovative solutions to address similar diagnostic gaps in childhood solid tumors and central nervous system cancers. While still in the early phases, DIVIA intends to ensure a practical and transformative solution for precise cancer diagnoses, no matter where a child lives.
Maximizing impact through health economics
Even when children are correctly diagnosed, access to effective treatment depends on whether health systems can afford and sustain the necessary medicines. Nancy Bolous, MD, MA, MSc, leads research within the DBS Unit focused on understanding the cost and cost-effectiveness of medical interventions. The work strives to answer key health economics questions about treatment choices, specifically comparing the cost and value of different therapeutic options.
“The goal is to identify treatments that meaningfully extend life while maintaining good health and well-being, ensuring that limited resources are invested in options that provide the greatest overall benefit to patients,” explained Bolous.
By comparing both the cost and outcomes of treatment, Bolous and her team can determine how much it costs to treat a patient with one option versus another and whether the additional expense leads to meaningful benefits. This work is important at the clinical level because it helps physicians choose between treatments by identifying which option provides the greatest benefit for specific patient populations. At the national level, this analysis informs health policy and budget planning, such as guiding prioritization of limited resources across diseases and treatment programs. This type of evidence also plays a critical role in advocacy, helping hospitals and foundations demonstrate how financial investments translate into lives saved, improved care or expanded access to treatment.
Two recently published studies in Blood and Cancer, focused on the costs of gene therapy and national cancer control plans for children, demonstrate the importance of the work being done. For example, one of the most expensive medicines is gene therapy for Hemophilia B. However, together with investigators from the St. Jude Good Manufacturing Practices Facility, the DBS Unit showed that the true cost to manufacture and deliver the treatment was less then 3% of the publicly available sticker price.
“The DBS unit initially started as an epidemiology program, but from the beginning, we knew we wanted it to be more than just counting cases,” explained Bhakta. “We wanted to turn data into action. That’s where the simulation and health economic components come in. We’ve designed it so that we can model how these numbers might flow through interventions, programs or ideas we develop globally, and use these data to understand the potential financial impact at the patient-level and at a global scale.”
Together, these layers ensure that funding decisions are informed by data and are aligned with the greatest possible impact for patients.
A hub for innovative solutions
The DBS Unit functions as a solutions-driven hub focused on tackling some of the biggest questions in global pediatric cancer. Through initiatives including SJCARES, the ARIA Guide and DIVIA, the unit develops tools, gathers data and conducts research to answer these questions, while also building a global community of collaborators.
In many ways, the unit acts as an innovation incubator, generating and refining projects until they are ready to be implemented more broadly. Each initiative that grows out of the unit remains connected to its mission of creating practical, scalable solutions for childhood cancer worldwide.






